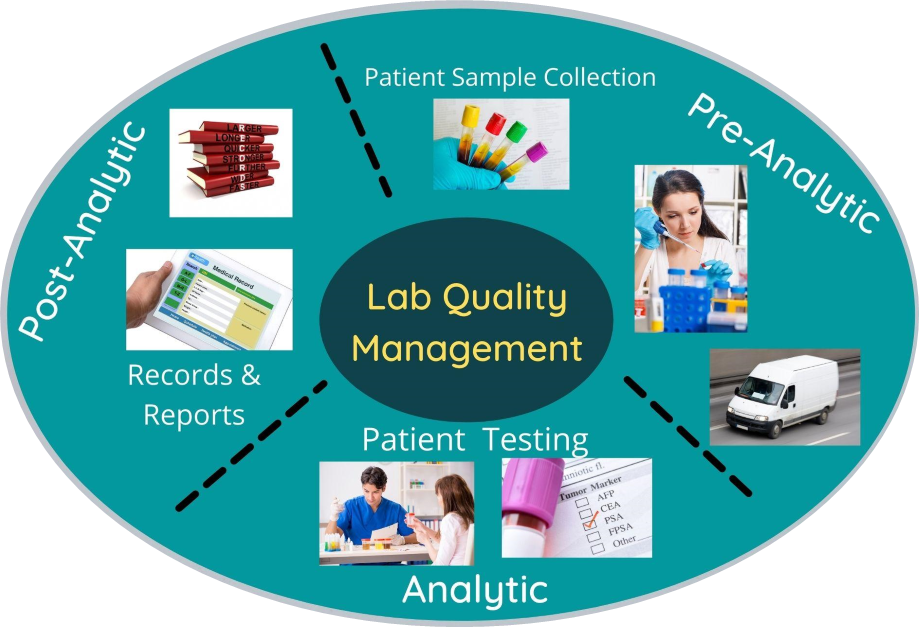
Quality Control Courses In Medical Laboratory
Quality Control Courses In Medical Laboratory - This course is targeted towards post graduate students of biochemistry, microbiology, and pathology. Master the use of statistical tools for quality control analysis. Understand the history of quality control and how it informs modern quality and statistical process controls. Utilize appropriate corrective actions for unacceptable quality control; Provide practical examples of qc in laboratory environments. Proper quality control helps ensure that reported results of patient laboratory testing are correct. Quality control refers to the statistical procedure which is used to monitor and evaluate analytical phase which is used to produce patient result. Laboratory quality can be defined as accuracy, reliability, and timeliness of the reported test results. Qc is an integral part of the total testing process and an essential element for producing accurate test results in the clinical laboratory. At career entry, the medical laboratory technician will be able to perform routine clinical laboratory tests (such as hematology, clinical chemistry, immunohematology, microbiology, serology/immunology, coagulation, molecular, and other emerging diagnostics) as the primary analyst making specimen oriented decisions on predetermined criteria, inc. Have greater knowledge of the supporting aspects and tools that can be utilised to design and implement quality assurance mechanisms. Laboratory mathematics covering performance of basic laboratory calculations including: Summarize the requirements for quality control documentation; Learn how the different elements of the quality management system—eg, internal audit, data analysis—play a role in identifying and controlling risk. Utilize appropriate corrective actions for unacceptable quality control; Compare methods for determining quality control acceptability; It describes a plan for managing the quality of your laboratory’s test results and services using the quality system essentials (qses), and introduces you to the individual courses that offer more detailed information on. Gain knowledge of international quality standards and regulatory requirements. Quality control is a comprehensive course in qc terminology, practices, statistics, and troubleshooting for the clinical laboratory. Quality control refers to the statistical procedure which is used to monitor and evaluate analytical phase which is used to produce patient result. Learn best practices for managing your risks, as well as practical tools that apply to all phases of the risk management process. A career in medical laboratory science combines the challenges of medicine, pathology,. Qc is an integral part of the total testing process and an essential element for producing accurate test results in the clinical laboratory. Have greater knowledge. Qc is an integral part of the total testing process and an essential element for producing accurate test results in the clinical laboratory. Decide when and how to perform quality control; At career entry, the medical laboratory technician will be able to perform routine clinical laboratory tests (such as hematology, clinical chemistry, immunohematology, microbiology, serology/immunology, coagulation, molecular, and other emerging. Learn best practices for managing your risks, as well as practical tools that apply to all phases of the risk management process. Elevate your medical lab quality management, assurance, and control expertise with our course on qa/qc, lqms principles, and the latest industry practices. Learn how the different elements of the quality management system—eg, internal audit, data analysis—play a role. Quality control (qc) is a system used to maintain a determined level of accuracy and precision. Decide when and how to perform quality control; Summarize the requirements for quality control documentation; Learn how the different elements of the quality management system—eg, internal audit, data analysis—play a role in identifying and controlling risk. This course is targeted towards post graduate students. Elevate your medical lab quality management, assurance, and control expertise with our course on qa/qc, lqms principles, and the latest industry practices. Serial and ratio dilutions, conversions of si and metric units, temperature conversions, statistical data for quality control and statistical analysis. The use of risk management is broadly applicable to all processes in the laboratory and can be used. Learn best practices for managing your risks, as well as practical tools that apply to all phases of the risk management process. This course is also useful for medical laboratory professional and technical staff working the medical laboratories. It describes a plan for managing the quality of your laboratory’s test results and services using the quality system essentials (qses), and. Qc is an integral part of the total testing process and an essential element for producing accurate test results in the clinical laboratory. Elevate your medical lab quality management, assurance, and control expertise with our course on qa/qc, lqms principles, and the latest industry practices. Designed for those who have little or no experience with quality control but need a. Summarize the requirements for quality control documentation; Quality control is a comprehensive course in qc terminology, practices, statistics, and troubleshooting for the clinical laboratory. Compare methods for determining quality control acceptability; It describes a plan for managing the quality of your laboratory’s test results and services using the quality system essentials (qses), and introduces you to the individual courses that. Learn how to implement quality control procedures in a laboratory setting. Its aim is to explain how qc works, explain what errors interpretive rules are designed to detect, and suggest appropriate investigations for qc failures. Choose quality assessment activities that are. Quality control is a comprehensive course in qc terminology, practices, statistics, and troubleshooting for the clinical laboratory. Quality control. The use of risk management is broadly applicable to all processes in the laboratory and can be used beyond the focus of qc. Understand the history of quality control and how it informs modern quality and statistical process controls. Have greater knowledge of the supporting aspects and tools that can be utilised to design and implement quality assurance mechanisms. Qc. This course is also useful for medical laboratory professional and technical staff working the medical laboratories. Quality control is a comprehensive course in qc terminology, practices, statistics, and troubleshooting for the clinical laboratory. The use of risk management is broadly applicable to all processes in the laboratory and can be used beyond the focus of qc. Serial and ratio dilutions, conversions of si and metric units, temperature conversions, statistical data for quality control and statistical analysis. Learn how to implement quality control procedures in a laboratory setting. Master the use of statistical tools for quality control analysis. Gain knowledge of international quality standards and regulatory requirements. Qc value assignment testing is critical in contributing meaningful data to support our diverse global qc customers and plays an integral part in the release of new assayed quality control lots. Choose quality assessment activities that are. This course is targeted towards post graduate students of biochemistry, microbiology, and pathology. The laboratory quality academy features interactive online learning modules, which provide an overview of quality as it applies to a laboratory setting, including a review of the quality system essentials (qses). Learn how the different elements of the quality management system—eg, internal audit, data analysis—play a role in identifying and controlling risk. Decide when and how to perform quality control; Quality control (qc) is a system used to maintain a determined level of accuracy and precision. This program focuses on the principles and applications of good statistical quality control (qc) practices. Compare methods for determining quality control acceptability;Quality Control vs Quality Assurance in Medical Laboratory
Laboratory Quality Control Assessment YouTube
Fundamentals of Laboratory Management OER Commons
Training Internal Quality Control Program in the Laboratory Training
Quality control in the medical laboratory
Quality Management in the Laboratory NATA
Part1 English Laboratory Quality Control Basics Biochemistry N
Quality control in the medical laboratory
Quality Control in Laboratory
The Purpose of a Laboratory Quality Assurance Program (QAP) LearnGxP
In This Course You Will Learn Principles Of Laboratory Management Including The Importance And Scope Of The Laws, Policies, And Regulations That Ensure Quality Medical Laboratory Practice In The U.s.
Quality Control Refers To The Statistical Procedure Which Is Used To Monitor And Evaluate Analytical Phase Which Is Used To Produce Patient Result.
Its Aim Is To Explain How Qc Works, Explain What Errors Interpretive Rules Are Designed To Detect, And Suggest Appropriate Investigations For Qc Failures.
Provide Practical Examples Of Qc In Laboratory Environments.
Related Post: